RNA therapy firm Avidity files $400m placement
pharmaphorum
MARCH 3, 2024
Avidity Biosciences files $400m private placement as it prepares for phase 3 trial of lead antibody oligonucleotide conjugate AOC 1001 in rare disease DM1
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

pharmaphorum
MARCH 3, 2024
Avidity Biosciences files $400m private placement as it prepares for phase 3 trial of lead antibody oligonucleotide conjugate AOC 1001 in rare disease DM1

pharmaphorum
SEPTEMBER 17, 2020
Eli Lilly has followed up supportive data from its Olumiant in COVID-19 with results of a study showing its antibody treatment could help prevent hospitalisation from the disease. The double-blind placebo-controlled antibody study involved patients with symptoms outside hospitals, who were treated with four different dose strengths.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.
Pharmaceutical Technology
JULY 15, 2022
Health Canada has granted approval for the usage of Moderna’s messenger RNA (mRNA) Covid-19 vaccine, Spikevax, in a 25µg two-dose regimen for active immunisation to prevent Covid-19 in children aged six months to five years. The KidCOVE trial was carried out at eight Canadian trial sites involving 414 children aged below five years.
Pharmaceutical Technology
JANUARY 19, 2023
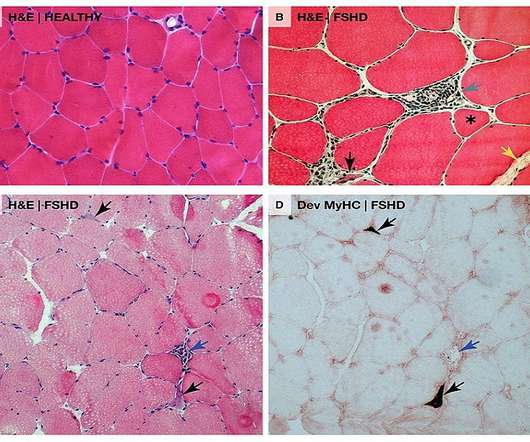
It comprises a monoclonal antibody (mAb) attached to the transferrin receptor 1 (TfR1) conjugated with a DUX4 mRNA that targets siRNA. We look forward to working collaboratively with the FDA to bring the first RNA therapy directly targeting DUX4 to patients as quickly as possible.”
Pharmaceutical Technology
FEBRUARY 3, 2023
Only a few weeks into the new year, the prospect of getting a successful advanced HIV vaccine shrank after the discontinuation of yet another late-stage trial. This marks the second time one of Janssen’s HIV vaccines failed after another showed disappointing results in the Phase IIb Imbokodo trial in August 2021.
Drug Discovery World
FEBRUARY 19, 2024
Replicate Bioscience has shared positive results from the Phase I trial of RBI-4000, its srRNA-based rabies vaccine. In this clinical trial, evaluating safety and immunogenicity of RBI-4000 , participants received one or two doses of srRNA vaccine at low doses (0.1mcg, 1mcg or 10mcg).

Pharmaceutical Technology
JUNE 6, 2023
According to Globaldata, it is involved in 1 clinical trial, which is ongoing. The therapeutic candidate consists of messenger RNA (mRNA) encoding for relaxin. It is developed based on messenger RNA expression and N2GL formulation platform. Buy the model here. The drug candidate is administered by intravenous route.

pharmaphorum
OCTOBER 16, 2020
Sanofi will shortly start human trials of a second coronavirus vaccine, developed with US biotech Translate Bio, after reporting that it stimulated antibodies against SARS-CoV-2 in preclinical testing. It started human testing of a protein subunit vaccine with partner GlaxoSmithKline last month. Chinese inactivated vaccine safe.

Drug Discovery World
MARCH 26, 2024
For example, progress made in developing gene mutant-specific inhibitors, antibody-drug conjugates (ADC), or cellular therapies. We were excited to unveil data showing that two distinct chemical series exhibit METTL1 inhibition in vitro at low nanomolar concentrations with minimal interference with other RNA and protein methyltransferases.
Pharmaceutical Technology
AUGUST 24, 2022
Mode rna has submitted an application to the US Food and Drug Administration (FDA) to obtain emergency use authorization (EUA) for mRNA-1273.222, its BA.4/BA.5 According to the findings, the trial of mRNA-1273.214 met all primary endpoints. At present, a Phase II/III trial of mRNA-1273.222 is progressing. 1 subvariant.

pharmaphorum
FEBRUARY 9, 2021
million ($22 million) in second-round financing as it prepares to start clinical trials of its lead drug candidate INT-1B3 for solid tumours. The start of dosing in the phase 1/1b trial of the drug has been delayed by the COVID-19 pandemic, but recruitment of the target 80 participants with advanced solid tumours is reported to be ongoing.

Delveinsight
SEPTEMBER 23, 2021
The biotech has evolved a T-cell receptor-like antibody to damage a selected immune checkpoint in oncology. 858 Therapeutics closes USD 60 Million series A round to drug RNA modulation. Now, they are ready for their next venture, which is launching RNA biotech 858 Therapeutics with a USD 60 million Series A fundraising.
Pharmaceutical Technology
JUNE 8, 2023
The company’s samRNA vector is based on a synthetic RNA molecule derived from a wild-type Venezuelan Equine Encephalitis Virus (VEEV) replicon with the goal of extending the duration and magnitude of immunogen expression to drive potent and durable immune responses. GlobalData is the parent company of Clinical Trials Arena.

XTalks
NOVEMBER 7, 2022
Gleich, MD, FACS, Senior Vice President, Medical Department, and Dr. Christopher Huth, PhD, Senior Clinical Trial Manager, Clinical Trial Management. Liquid Biopsy Use in Oncology Clinical Trials. Another method to isolate CTCs is by antibody targeting of white blood cells for removal, followed by collection of untagged CTCs.
Drug Discovery World
OCTOBER 11, 2023
The main presentations will be followed by a panel discussion on how these new approaches – e.g., engineered proteins, antibodies, micro RNA, nanocarriers, antibody drug conjugates – can help address unmet need in oncology.

Drug Discovery World
DECEMBER 21, 2023
Other biotechnologies coming to the fore this year were antibody-drug conjugates and CRISPR gene editing. Regulatory issues and diversity in clinical trials were also important in 2023, as well as the role that artificial intelligence tools like ChatGPT might play. DDW Multimedia Editor Megan Thomas explored three trends in the market.

Delveinsight
AUGUST 26, 2021
Shape’s RNA editing technologies can modify the RNA sequence, which makes the body’s protein building blocks. This system is designed to deliver RNA editing technology or other payloads directly to particular body areas, such as the nervous system or muscle. and Leila Zegna, director of the Kabuki Syndrome Foundation.
The Pharma Data
OCTOBER 25, 2020
The messenger RNA-based vaccine, which is designed to block replication of the coronavirus, was shown to induce neutralizing antibodies and activate T-cells in hamsters and mice. The vaccine is currently being evaluated in phase 1 and 2 human clinical trials. Source link.
The Pharma Data
MARCH 11, 2021
Clinical trial to assess safety, immune response and reactogenicity, after preclinical data showed high neutralizing antibody levels. The Companies expect interim results from this trial in the third quarter of 2021. About the Phase 1/2 clinical trial. About previously-published preclinical results.
The Pharma Data
DECEMBER 8, 2020
8, 2020 — AstraZeneca’s COVID-19 vaccine is safe and effective, new data from late-stage trials shows. The interim findings are from phase 3 trials in the United Kingdom and Brazil that included more than 11,600 participants. TUESDAY, Dec. 8 in The Lancet. New data posted Tuesday on the U.S. © 2020 HealthDay.
Pharmaceutical Technology
JULY 5, 2022
On leaving the liver cells, the virus hijacks bits of cell membrane and conceals itself from antibodies that would have isolated the virus before it travelled far through the bloodstream. In addition, the scientists later found that the HAV needs TENT4A/B for its replication.

XTalks
NOVEMBER 30, 2023
Over 26 weeks of Sunlenca combined with other antiretroviral drugs, 81 percent of participants achieved HIV RNA suppression, reaching levels low enough to be considered undetectable. Both trials concluded with maintained virologic suppression, and no clinically significant change from baseline in CD4+ cell counts was observed.

pharmaphorum
JANUARY 7, 2022
NI006 is an antibody that binds to the misfolded forms of the transthyretin protein that form the fibrils, and cause them to be cleared away by immune cells like macrophages. billion on the first nine months of 2021, well ahead of Pfizer’s own forecasts. Tafamidis is thought to work by stabilising the misfolded proteins.

Drug Discovery World
AUGUST 4, 2023
The top stories: Promontory Therapeutics expands Phase II mCRPC trial Promontory Therapeutics has treated its first patients in France as part of the ongoing Phase II clinical trial of PT-112 in patients with late-stage metastatic castration-resistant prostate cancer (mCRPC).

The Pharma Data
JANUARY 31, 2021
Adjuvanted S-Trimer COVID-19 vaccine candidates demonstrated favorable safety and tolerability profiles and strong neutralizing immune responses in a phase 1 trial. Clover plans to initiate a global phase 2/3 trial in the first half of 2021 with an interim analysis for vaccine efficacy potentially in the middle of 2021.

pharmaphorum
SEPTEMBER 16, 2020
Both of these vaccines are based on RNA and in phase 3 and phase 1 testing, respectively. Earlier clinical data has shown that the vaccine can stimulate neutralising antibodies against SARS-CoV-2 and was well-tolerated with a transient mild to moderate fever being the main side effect seen in less than 20% of recipients.
The Pharma Data
JANUARY 20, 2021
-based Gritstone Oncology is developing a novel COVID-19 vaccine based on adenovirus and messenger-RNA technology designed to combat future variants of the coronavirus. Louis University. . Source link.

The Pharma Data
NOVEMBER 16, 2020
November 16, 2020 — An independent data and safety monitoring board (DSMB) overseeing the Phase 3 trial of the investigational COVID-19 vaccine known as mRNA-1273 reviewed trial data and shared its interim analysis with the trial oversight group on Nov. 37% of trial volunteers are from racial and ethnic minorities.
pharmaphorum
DECEMBER 1, 2020
The companies initially filed the nonclinical data and other technical data, followed by emerging clinical trial results. In the trial, BNT162b2 also showed an overall favourable tolerability with no safety concerns reported to date. The first primary objective analysis was based on 170 confirmed cases of COVID-19.
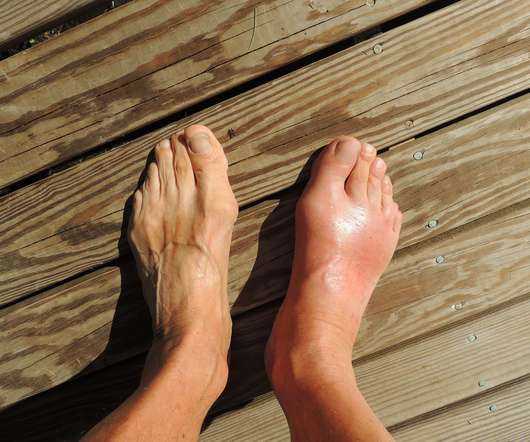
XTalks
JULY 14, 2022
Krystexxa’s expanded approval was based on data from the randomized, controlled MIRROR trial involving adults with uncontrolled gout. Results from the trial showed “significant improvement and sustained response” to Krystexxa with methotrexate (a chemotherapy drug and immune suppressant) compared to Krystexxa alone.

Roots Analysis
OCTOBER 13, 2023
As a result, in late nineties, the conjugation of drug with biological entities such as antibodies emerged as a possible solution for targeting earlier undruggable entities. However, it was not until 2000 when the first drug conjugate in the form of antibody drug conjugate (ADC) was approved by the USFDA.

Roots Analysis
DECEMBER 12, 2023
As a result, innovators came up with the idea of magic bullets, antibody drug conjugates (ADCs). It is comprised of three components, namely antibody, drug and linker. The antibody is conjugated to the drug via a linker molecule. The antibody is conjugated to the drug via a linker molecule. Components of an ADC?
pharmaphorum
NOVEMBER 23, 2020
AstraZeneca is preparing to file its COVID-19 vaccine with regulators after phase 3 trial results showed it is up to 90% effective. Results from trials conducted in the UK and Brazil showed an efficacy of 90% in 2,741 patients receiving the vaccine as a half dose, followed by a full dose at least one month apart.

The Pharma Data
JANUARY 31, 2021
Clover plans to initiate a global Phase 2/3 efficacy trial of its protein-based S-Trimer COVID-19 vaccine candidate adjuvanted with Dynavax’s CpG 1018 plus alum in the first half of 2021 with an interim analysis for vaccine efficacy potentially in the middle of 2021. EMERYVILLE, Calif. and CHENGDU, China , Feb.

pharmaphorum
OCTOBER 6, 2020
In the case of a rolling review, the EMA evaluates trial data as they become available before a complete filing is submitted. These studies suggest BNT162b2 triggers the production of neutralising antibodies and T-cells that target SARS-CoV-2, the coronavirus that causes COVID-19.

pharmaphorum
DECEMBER 23, 2021
The first-in-class, small interfering RNA (siRNA) therapy for cholesterol lowering inhibiting PCSSK9 – the same target as Amgen’s Repatha (evolocumab) and Sanofi/Regeneron’s Praluent (alirocumab) – but after a lead-in period is dosed only twice a year rather than every month.

World of DTC Marketing
MARCH 16, 2021
Do I have a responsibility to share — on every occasion, random and formal, to family, friends, and strangers alike — my knowledge on the vaccines, monoclonal antibodies, the latest news from the Lancet or JAMA, why RNA is not voodoo, how Phase 4 safety trials are happening as I write…The answer is simple: yes.
pharmaphorum
SEPTEMBER 22, 2022
Le Vert says Osivax’s technology differs, however, as it attacks the virus’ T-cells, making the vaccine more effective in the long term, and that clinical trials have shown promise. When exposed to a virus, one’s immune system develops antibodies in response to foreign antigens, with or without a vaccine.

pharmaphorum
APRIL 5, 2022
AstraZeneca’s take on the PCSK9 inhibitor – an antisense drug licensed from Ionis six years ago – has cleared a proof-of-concept trial in high-risk patients with high blood cholesterol, but it remains to be seen where it will fit into the increasingly busy category.
pharmaphorum
JANUARY 5, 2022
A COVID-19 vaccine that could work against multiple variants of the coronavirus – developed by US biotech Gritstone bio – has generated encouraging immune response data in its first clinical trial.

BioSpace
NOVEMBER 20, 2022
Former Theranos CEO Elizabeth Holmes will serve 11 years in prison on four counts of fraud, according to her sentencing by a federal judge in a California courtroom on Friday afternoon.
pharmaphorum
NOVEMBER 17, 2020
The vaccine developed in partnership with Germany’s BioNTech has been shown to be 90% effective in clinical trials. The company has picked four US states – Rhode Island, Texas, New Mexico and Tennessee, to trial a delivery programme that aims to overcome this challenge.

The Pharma Data
AUGUST 16, 2021
Better activation of innate and adaptive immune responses was achieved with CV2CoV, resulting in faster response onset, higher titers of antibodies, and stronger memory B and T cell activation as compared to the first-generation candidate, CVnCoV. The first clinical trial of CV2CoV is expected to start in Q4 2021.

pharmaphorum
NOVEMBER 21, 2022
Bomedemstat (IMG-7289) – an oral lysine-specific demethylase 1 (LSD1) inhibitor – is in multiple phase 2 trials for the treatment of indications including essential thrombocythemia (ET), myelofibrosis (MF), and polycythemia vera (PV). The acquisition is expected to close in the first quarter of 2023.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content