Novo Nordisk agrees $1.1bn takeover of RNA biotech Cardior
pharmaphorum
MARCH 25, 2024
billion, adding expertise in RNA therapeutics and a heart failure therapy in mid-stage trials. Novo Nordisk has agreed to buy Cardior Pharmaceuticals for $1.1
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

pharmaphorum
MARCH 25, 2024
billion, adding expertise in RNA therapeutics and a heart failure therapy in mid-stage trials. Novo Nordisk has agreed to buy Cardior Pharmaceuticals for $1.1

pharmaphorum
MAY 2, 2024
A personalised vaccine for the aggressive brain cancer glioblastoma developed has shown encouraging signs of efficacy in its first human trial.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Pharmaceutical Technology
JUNE 16, 2023
from Flanders Innovation & Entrepreneurship (VLAIO) to further advance its oncology portfolio targeting RNA. It will also support the firm’s preclinical effort on the long non-coding RNA (LncRNA) programme, FTX-001, that targets MALAT-1. Flamingo Therapeutics has received a research grant of €1.7m
Pharmaceutical Technology
MARCH 1, 2023
Further, the acceptance of new mRNA vaccines has rejuvenated activity within previously established categories of RNA therapeutics including lifesaving antisense technologies. These include antisense oligonucleotides (ASO), RNA interference (RNAi), and RNA aptamers.
Pharmaceutical Technology
MAY 11, 2023
The vaccine, developed by BioNTech, led to half of the patients with pancreatic cancer in the Phase I trial remaining cancer-free 18 months later. A global randomised follow-up trial is planned. In 2016, BioNTech and Genentech, a part of Roche signed an agreement to develop personalised mRNA therapies in oncology.

AuroBlog - Aurous Healthcare Clinical Trials blog
NOVEMBER 1, 2023
The Drug Controller General of India (DCGI) has added in-vitro diagnostic (IVD) medical devices including those for diagnosis of Covid-19, ribonucleic acid (RNA) and deoxyribonucleic acid (DNA) extraction kits, among others into the Class C risk category under the Medical Devices Rules (MDR), 2017.

Pharmaceutical Technology
JULY 29, 2022
Last week, CAMP4 Therapeutics announced the close of a $100 million Series B round , which will be used to advance their regulatory RNA (regRNA)-focused programs. CAMP4’s CSO David Bumcrot PhD tells Pharmaceutical Technology that the company plans to see clinical trials go forward for their urea cycle disorder programs late next year.

pharmaphorum
AUGUST 16, 2022
Merck & Co has ramped up its involvement in the RNA category, partnering with US biotech Orna Therapeutics in a deal valued at up to $3.5 Now, Merck has made its own play, partnering with Orna on its proprietary ‘oRNA’ technology, which stands for circular RNA. billion, including $150 million upfront.

Bio Pharma Dive
FEBRUARY 10, 2023
The pharma and partner BioNTech have kicked off the first human trial of a messenger RNA shot for shingles, believing there’s room to clear the high bar set by GSK’s rival vaccine.

XTalks
SEPTEMBER 13, 2021
With RNA therapies being the next hot thing in genetic medicine, Eli Lilly is joining the RNA editing race by partnering with Netherlands-based ProQR Therapeutics NV (Nasdaq: PRQR), a biotech company developing RNA-based therapies for rare genetic diseases with a focus on blinding disorders of the retina.

pharmaphorum
MARCH 3, 2024
Avidity Biosciences files $400m private placement as it prepares for phase 3 trial of lead antibody oligonucleotide conjugate AOC 1001 in rare disease DM1

Bio Pharma Dive
MAY 19, 2023
The biotech, best known for its vaccine research, said its clinical trial is the first to report results of a messenger RNA therapeutic designed for intracellular protein replacement.

Roots Analysis
NOVEMBER 6, 2023
RNA-based therapeutics have completely revolutionized the healthcare segment, greatly influencing the study and treatment of human diseases by conferring precise targeting ability to therapeutic modalities. The below figure presents the distribution of next generation RNA-based therapeutics and RNA-based vaccines, based on type of molecule.

Drug Discovery World
APRIL 11, 2023
US-based start-up Atomic AI is using artificial intelligence (AI) to design more intelligent RNA-targeted and RNA-based medicines. The technology can also generate synthetic data sets, or make human trials more efficient, by identifying the most suitable patients, predicting outcomes and optimising dosages and regimens of drugs.
Drug Discovery World
DECEMBER 13, 2022
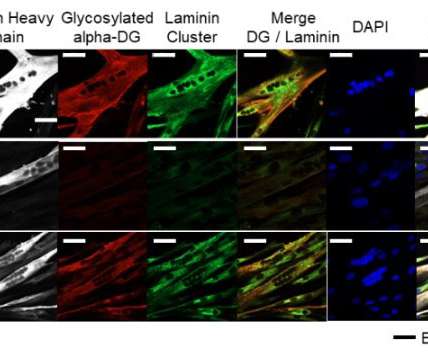
Japanese researchers propose using RNA interference for correcting a mistake in the genetic code of patients with Fukuyama muscular dystrophy. . Using RNA interference, researchers restored the normal biological function of FKTN in patient-derived cells, offering hope for a new therapy. . Modifying RNA function.
Drug Discovery World
AUGUST 1, 2023
Investigators at the Icahn School of Medicine at Mount Sinai have designed an RNA-based strategy to activate dendritic cells that eradicated tumours and prevented their recurrence in mouse models of melanoma. However, these have not been as successful in clinical trials as hoped.
STAT News
JANUARY 9, 2023
An experimental RNA treatment reduced liver scarring in half of patients with an inherited disease called alpha-1 antitrypsin deficiency, or AATD, according to results from a mid-stage clinical trial reported Monday by its maker Arrowhead Pharmaceuticals.

XTalks
MARCH 21, 2022
Credited as a pioneering RNA interference (RNAi) therapeutics company, Alnylam Pharmaceuticals is taking Pfizer and Moderna to court, claiming that the companies’ use of the lipid nanoparticle (LNP)-based RNA delivery technology in their mRNA COVID-19 vaccines infringes on a patented technology.

Drug Discovery World
DECEMBER 5, 2022
Based on Nakhoda and Olszanski, 2022 (Addressing Recent Failures in Immuno-Oncology Trials to Guide Novel Immunotherapeutic Treatment Strategies), lack of an optimal biomarker is one of the main challenges to clinical trial success. What you will learn: Overview: Relevance of single-cell RNA sequencing in your drug discovery toolkit.

Drug Discovery World
DECEMBER 16, 2022
The investment allows the company to focus on discovering and developing novel small molecule therapies targeting RNA modifying enzymes (RMEs) for oncology and other diseases. . The post Biotech company raises $30M to target RNA modifying enzymes appeared first on Drug Discovery World (DDW).

Fierce Pharma
APRIL 22, 2024
As a closely watched readout from Alnylam Pharmaceuticals' Amvuttra in the rare heart disease transthyretin amyloid cardiomyopathy (ATTR-CM) is expected in a few weeks, experts suggested the RNA si | As an all-important readout from Alnylam’s Amvuttra in the rare heart disease ATTR-CM is expected in a few weeks, experts suggested that the RNA (..)
Fierce Pharma
FEBRUARY 15, 2024
Changing a clinical trial’s statistical analysis plan on the cusp of a readout? Changing a clinical trial’s statistical analysis plan on the cusp of a readout? That’s exactly what Alnylam just did for a closely watched study of its next-generation RNA interference therapy Amvuttra in a rare heart disease.

XTalks
JANUARY 8, 2024
Whether it’s for a treatment for a chronic ambulatory condition, precision medicine or cell and gene therapy, there is a massive uptick in clinical trial complexity. It’s important to make sure that with this increase in clinical trial complexity, we don’t make our trials overly burdensome to sites or patients,” says Markham.

BioTech 365
JANUARY 17, 2022
Brazil’s SENAI CIMATEC Doses First Healthy Volunteers in Phase 1 trial of HDT Bio’s RNA COVID-19 Vaccine Brazil’s SENAI CIMATEC Doses First Healthy Volunteers in Phase 1 trial of HDT Bio’s RNA COVID-19 Vaccine HDT Bio’s RNA-based vaccine offers simpler … Continue reading →

BioTech 365
AUGUST 31, 2021
HDT Bio Corp and Gennova Complete Phase 1 Trial of COVID-19 RNA Vaccine in India HDT Bio Corp and Gennova Complete Phase 1 Trial of COVID-19 RNA Vaccine in India Vaccine Would Be India’s First Indigenous RNA Vaccine, Exemplifying HDT … Continue reading →

pharmaphorum
JANUARY 6, 2022
Roivant is the latest pharma group to take a position in the emerging field of therapies targeted at RNA splicing, licensing exclusive rights to a drug developed by a unit of Japanese drugmaker Eisai. . The post Roivant joins RNA splicing push with Eisai deal appeared first on.

Pharmaceutical Technology
MARCH 28, 2023
There is much interest in the industry around RNA-based therapeutics as their utilisation in indications beyond Covid-19 come into focus. In December 2022, BioNTech initiated a Phase I clinical trial of BNT163 – an HSV vaccine candidate. In 2022, the company also started five first-in-human clinical trials from its oncology programme.
Drug Discovery World
DECEMBER 1, 2022
Based on Nakhoda and Olszanski, 2022 (Addressing Recent Failures in Immuno-Oncology Trials to Guide Novel Immunotherapeutic Treatment Strategies), lack of an optimal biomarker is one of the main challenges to clinical trial success. What you will learn: Overview: Relevance of single-cell RNA sequencing in your drug discovery toolkit.

pharmaphorum
JULY 20, 2022
Armed with a $100 million second-round financing, CAMP4 Therapeutics is preparing to start the first clinical trial of a drug targeting regulatory RNA (regRNA) molecules that can be used to fine-tune the expression of genes. ” The post CAMP4 raises $100m to take lead RNA drugs into clinic appeared first on.

BioTech 365
JULY 6, 2021
Biotechnology, Pharma and Biopharma News – Research – Science – Lifescience ://Biotech-Biopharma-Pharma: Lilly invests in MiNA to fund trials of protein-upregulating RNA.Lilly invests in MiNA to fund trials of protein-upregulating RNA ntaylor Tue, 07/06/2021 – 07:58 from FierceBiotech: Biotech … Continue reading → (..)

BioTech 365
DECEMBER 13, 2021
HDT Bio Partner Quratis Doses First Healthy Volunteers in Phase 1 Trial of HDT Bio’s RNA COVID-19 Vaccine in South Korea HDT Bio Partner Quratis Doses First Healthy Volunteers in Phase 1 Trial of HDT Bio’s RNA COVID-19 Vaccine in … Continue reading →

Bio Pharma Dive
AUGUST 11, 2022
The company will wind down five trials involving two drug candidates and lay off more staff, prioritizing instead its RNA-editing technology.

STAT News
JANUARY 30, 2023
It is now possible to treat diseases with gene therapy, antisense oligonucleotides, messenger RNA (mRNA), noncoding RNA (known as small interfering RNA, or siRNA), and other gene-based modalities. New ways of conducting clinical trials have also emerged. The human genome was sequenced in 2003. Read the rest…

pharmaphorum
DECEMBER 14, 2022
The two partners have revealed the first clinical data from a phase 2b trial of a personalised vaccine consisting of 34 mRNAs, each targeting mutations – identified by gene sequencing – that are thought to be driving a patient’s cancer. Access to sequencing could, however, be a major obstacle at the population level.

AuroBlog - Aurous Healthcare Clinical Trials blog
OCTOBER 16, 2022
The husband-and-wife team who co-founded BioNTech, the biotechnology company that partnered with Pfizer to develop an effective messenger-RNA (mRNA) shot against COVID-19, has predicted that a cancer vaccine could be widely available within the next decade.
Pharmaceutical Commerce
FEBRUARY 12, 2024
In clinical trials, bepirovirsen was found to recognize the RNA used by hepatitis B virus to replicate in infected liver cells.
Drug Discovery World
MAY 18, 2023
A clinical trial of an experimental universal influenza vaccine developed by researchers at the National Institute of Allergy and Infectious Diseases’ (NIAID) Vaccine Research Center (VRC) has begun enrolling volunteers at Duke University in Durham, North Carolina. The trial will enrol up to 50 healthy volunteers aged 18 to 49.
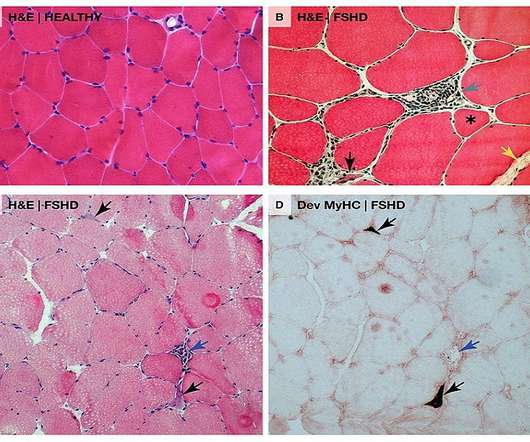
Pharmaceutical Technology
JANUARY 19, 2023
“We look forward to working collaboratively with the FDA to bring the first RNA therapy directly targeting DUX4 to patients as quickly as possible.” AOC 1020 is currently being studied in the double-blind, randomised, placebo-controlled Phase I/II FORTITUDE clinical trial in nearly 70 FSHD adult patients.

Pharmaceutical Technology
NOVEMBER 10, 2022
Olpasiran is entering a Phase III clinical trial to evaluate if the siRNA therapy can reduce cardiovascular event risk in atherosclerotic cardiovascular disease patients with increased Lp(a) levels. . The post Royalty Pharma acquires royalty interest in olpasiran for $250m appeared first on Pharmaceutical Technology.
Pharmaceutical Technology
MAY 19, 2023
Myeloid Therapeutics has raised $73m to support the continued clinical development of its lead cell therapy programme, MT-101, in Phase I/II trials for T cell lymphoma. The financing will help to fast-track the development of other in vivo programming candidates into clinical trials.

The Pharma Data
DECEMBER 10, 2020
Moderna has begun dosing adolescents in a phase 2/3 trial evaluating messenger RNA-based vaccine candidate mRNA-1273 and plans to enroll 3,000 U.S. participants aged 12 to 17. Source link.
Pharmaceutical Technology
OCTOBER 14, 2022
In 2016, the companies entered a strategic partnership to develop novel messenger RNA (mRNA) based PCVs. It is currently being assessed in combination with Merck’s anti-PD-1 therapy, Keytruda, as an adjuvant treatment for high-risk melanoma patients in a Phase II clinical trial being conducted by Moderna.

Drug Discovery World
JULY 19, 2022
An early-stage clinical trial has been launched in the US investigating a vaccine against Nipah virus infection. . The clinical trial has been launched by the National Institute of Allergy and Infectious Diseases (NIAID) to evaluate an investigational vaccine to prevent infection with Nipah virus.
Drug Discovery World
FEBRUARY 19, 2024
Replicate Bioscience has shared positive results from the Phase I trial of RBI-4000, its srRNA-based rabies vaccine. In this clinical trial, evaluating safety and immunogenicity of RBI-4000 , participants received one or two doses of srRNA vaccine at low doses (0.1mcg, 1mcg or 10mcg).
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content