Merck builds in cell, gene therapies with Mirus Bio deal
pharmaphorum
MAY 23, 2024
Merck KGaA has agreed to a $600m deal to buy Mirus Bio, a specialist in transfection reagents used in the production of cell and gene therapies.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

pharmaphorum
MAY 23, 2024
Merck KGaA has agreed to a $600m deal to buy Mirus Bio, a specialist in transfection reagents used in the production of cell and gene therapies.

BioPharma Reporter
AUGUST 14, 2023
Teknova, a producer of life science reagents, has opened a new GMP-certified production facility which it claims will enable bioprocessing and gene therapy companies to get into the clinic faster.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

BioPharma Reporter
FEBRUARY 22, 2024
MIP Discovery, a developer of non-biological affinity reagents designed to accelerate the production of cell and gene therapies, has closed a Â7 million Series A financing round, led by Mercia Ventures.

Pharmaceutical Technology
APRIL 3, 2023
The deal will see Polyplus join the German life science group’s portfolio allowing the latter to leverage expertise in transfection reagents and plasmid DNA for gene therapy. Polyplus, based in Strasbourg, France, produces key components in the production of viral vectors used in cell and gene therapies.

Drug Discovery World
FEBRUARY 21, 2024
Using MIPs’ sophisticated and highly scalable synthetic chemistry approach means they are well positioned to benefit from this tremendous high growth opportunity.” Liz Klein, Investment Director at Calculus, added: “MIP Discovery’s synthetic affinity reagents hold huge potential to overcome bottlenecks in CGT development and production.

BioTech 365
SEPTEMBER 20, 2021
Global Gene Expression Market Size, Share & Trends Analysis 2021-2028 by Product (DNA Chip/Microarray, Kits & Reagents), Technique (Promoter Analysis, RNA Exp.) – ResearchAndMarkets.com Global Gene Expression Market Size, Share & Trends Analysis 2021-2028 by Product (DNA Chip/Microarray, Kits & … Continue reading → (..)
BioPharma Reporter
AUGUST 18, 2021
Recombinant adeno-associated viruses (rAAVs) are becoming the vector of choice for delivering gene therapies, says the French firm.

Drug Discovery World
MAY 25, 2023
Several new products for the life sciences sector have been launched over the last month, from miniature semiconductor chip (MSC) technology to a new inhalation testing service. The platform works with the SYNTAX Hi-Fidelity reagent kits for synthesising longer oligos. DDW provides an update.

Camargo
MAY 2, 2021
Advances in scientific knowledge and growth in the cell and gene therapy space have led to a new and exciting era of medicine for patients, as well as a new motivation for regulators to provide clear, efficient pathways for product developers. Background: The Advancement of Cell and Gene Therapies.
Drug Discovery World
MAY 7, 2024
Where the bottlenecks of achieving higher cell density and higher titers were once thought to be issues of the past, hurdles have now shifted to achieving speed and scale in mAb production without sacrificing quality. DC: Antibodies have and remain a large product class within the biopharmaceutical market.

BioPharma Reporter
JULY 5, 2021
Sartorius is strengthening its product portfolio for cell and gene therapies with the acquisition of a majority stake in German reagent manufacturer CellGenix.
Advarra
NOVEMBER 29, 2022
The field of cell and gene therapies (CGT) is constantly evolving, and there has been significant progress in this area of research. Recent Problems in Cell and Gene Therapy Development. Dealing with confusing regulatory guidelines is one of the top obstacles researchers face as they develop CGT products 1.
Drug Discovery World
AUGUST 11, 2023
GenScript Biotech and T-MAXIMUM Biotech have formed a strategic collaboration to develop T-MAXIMUM’s CAR-T cell therapy using GenScript’s CRISPR nucleic acid reagents. GenScript will provide various CRISPR reagents to support the development of the universal CAR-T products from discovery to commercialisation.
Drug Discovery World
SEPTEMBER 25, 2023
CRISPR gene editing companies Integrated DNA Technologies (IDT) and Aldevron have inked a new global distribution agreement to expand CRISPR products for cell and gene therapy customers.
Drug Discovery World
MAY 16, 2023
What are the global innovations in cell and gene therapy? Skills gap Bruce Levine, International Society for Cell & Gene Therapy (ISCT) Immediate Past President and Chair, Strategic Advisory Council, says a critical challenge that has become apparent for the CGT sector is the need for workforce development and training resources.

FDA Law Blog
JANUARY 4, 2023
Brevig, Senior Regulatory Device and Biologics Expert — On December 7, 2022, FDA’s Center for Biologics Evaluation and Research (CBER) and the Office of Tissues and Advanced Therapies (OTAT) held a town hall to answer questions related to cell therapy and tissue-engineered products chemistry, manufacturing, and controls (CMC).

Roots Analysis
FEBRUARY 21, 2022
Since the introduction of a gene sequencing method by Frederick Sanger in 1977, the field of genomic data collection and analysis has evolved significantly. Moreover, the manual protocols require extensive manipulation, costly reagents and long duration of skilled genomic library production. Product Competitiveness Analysis.

Drug Discovery World
JANUARY 30, 2024
Viral vector manufacturing At the Advanced Therapies Awards ceremony, Dr Luigi Naldini, Director of the San Raffaele Telethon Institute for Gene Therapy, was awarded a Lifetime Achievement Award in recognition of ‘extraordinary contributions to gene therapy’, a field he has helped shape through his pioneering work on lentiviral vectors.

The Pharma Data
MAY 17, 2021
Academic centers should establish risk management programs to successfully commercialize cell and gene therapy products and to be a viable partner with contract research organizations (CROs) and manufacturers in developing these therapies. . Uharek is a hematologist and cell and gene therapy specialist at Berlin’s Charité Hospital.

pharmaphorum
DECEMBER 16, 2022
Maishman brings a wealth of experience to the cell and gene therapy-focused company, having worked at GlaxoSmithKline, Pfizer, Valneva, Alexion, and Recipharm in various leadership roles. Reagents supplier The Native Antigen Company has hired a new business development manager, Richard White , and senior product manager, Pardeep Sharda.

Roots Analysis
FEBRUARY 23, 2022
Over the past few years, several companies have started offering a diverse range of genome sequencing products and services using various second and third generation sequencing technologies. Next Generation Sequencing (NGS) Library Preparation Kits. NGS Library Preparation Kits Market.
Drug Discovery World
JANUARY 10, 2023
For over 50 years, Bethyl Laboratories (now a Fortis Life Sciences brand) has been dedicated to improving lives by supporting scientific discovery through qualified antibody products and custom services. This pillar uses gene knockout or knockdown to reduce the levels of target protein available for detection. Our areas of expertise.

The Pharma Data
OCTOBER 14, 2020
Bio-Techne’s Woburn facility produces Cloudz Cell Activation Kits, a magnetic-free bead-based technology for the robust and reliable expansion of immune cells, including T cells and NK cells, representing a pivotal component of the cell and gene therapy workflow. ” About Bio-Techne Corporation (NASDAQ: TECH). .

Pharmaceutical Technology
FEBRUARY 3, 2023
With scientists fervently developing mRNA vaccines, nucleic acid therapeutics, and viral vector-based gene therapies, clinicians are set to have a growing number of tools available to treat a wide range of conditions, from infectious diseases to genetic disorders and more.

The Pharma Data
JANUARY 24, 2021
. “We are excited to welcome Chris, a highly regarded drug developer and team builder who brings to Notch great depth of experience and expertise in development of cell therapies, gene editing, and cell engineering spanning discovery through IND,” said David Main , President and Chief Executive Officer of Notch.

Roots Analysis
OCTOBER 16, 2023
DNA and Gene Cloning involves the isolation of a DNA sequence of any species ( often a gene ) and its insertion into a vector to enable growth without any alteration in the original DNA sequence. There are some motifs, such as hairpin loops which hinder the usability of a gene.

Drug Discovery World
OCTOBER 13, 2022
Another productive panel included Science Solutions Recruitment – Futureproof your career in Drug Discovery , featuring Paul Mears from Science Solutions Recruitment and Andrew Ratcliffe from Domainex. . Speakers included Ms Hanadi Jabado, Mr Tom Fleming, Dr Michael Salako and Dr Margaret Anne Craig. Looking forward .

Drug Discovery World
MARCH 13, 2023
The cell and gene therapy (CGT) landscape has grown significantly in the past year. One example: Zynteglo, a newly approved gene therapy priced at a record-breaking $2.8M Facilities for CGT development require a high level of maintenance, specialised equipment, and a fill/finish facility to produce sterile drug products.

Drug Discovery World
DECEMBER 23, 2022
GenScript USA has expanded its global current Good Manufacturing Practices (cGMP) single-guide RNA (sgRNA) manufacturing capabilities to meet the growing demand for cell and gene therapies. . The 400,000 square-foot cGMP facility enables gene and cell therapy R&D programmes to advance IND filings and clinical trials.
pharmaphorum
FEBRUARY 3, 2021
Dr Jennifer Harbottle, senior scientist in the R&D Base Editing team of PerkinElmer’s Horizon Discovery business, looks at progress made in the realms of biotechnology and next-generation diagnostics, vaccines and therapeutics, including the application of CRISPR-Cas9 gene editing in developing and refining cell therapies.
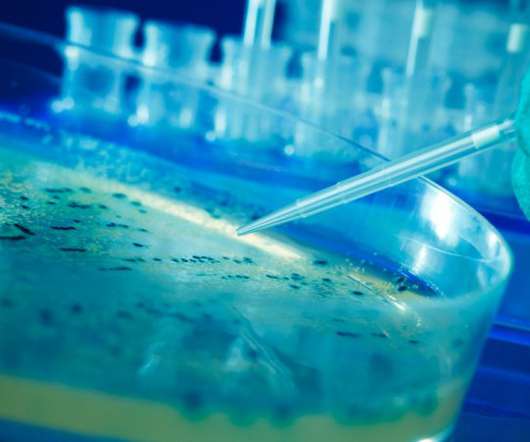
Drug Discovery World
JANUARY 19, 2023
This paid-for advertorial by Catalent appeared in DDW Volume 24 – Issue 1, Winter 2022/2023 One significant challenge in the manufacturing of cell and gene therapies is the production of high-quality plasmid DNA (pDNA). Figure 1: The integrated development pathway for cell and gene therapies.

The Pharma Data
NOVEMBER 30, 2020
Vivlion holds an exclusive license to Goethe University of Frankfurt’s proprietary 3Cs technology for the production of next generation 3Cs CRISPR/Cas gRNA libraries. The license from ERS Genomics now enables Vivlion to offer both R&D reagents and screening services to its customers worldwide. “It CEO of Vivlion.

Drug Discovery World
JUNE 14, 2023
BCA members also operate the BCA Advanced Therapies Network which is a diverse, fully integrated portfolio of blood, cell, gene and tissue materials that support the development and delivery of advanced therapies. million blood units annually, or 50% of the nation’s blood supply.

Drug Discovery World
FEBRUARY 8, 2023
Svea Cheeseman , Refeyn, explains why better efficiency is needed to advance the production of viral vectors for use in gene therapies. Adeno-associated viruses (AAVs) are a promising, widely used vector for delivering gene therapies. Data were provided by the Cell and Gene Therapy Catapult (London, UK).

Drug Discovery World
OCTOBER 12, 2022
This progress includes the expansion of genomic sciences and the ability to design cell bioprocessing platforms for biotherapeutics production that can be used to address previously unmet medical needs. However, obstacles to large-scale production and usage of these biotherapeutics are hampering progress. The glycan database .

Drug Discovery World
NOVEMBER 30, 2023
coli expression systems and is using high-throughput expression pipelines to screen constructs for optimal expression to generate protein and cellular reagents. It was presented by Lei Shi, PhD, Senior Vice President, R&D, Biointron Biological, titled: ‘Accelerating early discovery through HTP and high-speed antibody production’.

The Pharma Data
DECEMBER 2, 2020
ERS Genomics – Ireland’s ERS Genomics Limited, and Germany’s Vivlion GmbH, announced a non-exclusive license agreement granting Vivlion access to ERS Genomics’ CRISPR/Cas9 patent portfolio, to enhance Vivlion’s gene editing reagents and screening services. Financial details of the agreement were not disclosed. in Mainland China.

Drug Discovery World
JULY 18, 2023
Since its discovery in 2012, CRISPR (Clustered Regularly Interspaced Short Palindromic Repeats) technology has revolutionised the biomedicine and cell and gene therapy fields, providing a versatile tool for precise and efficient genome editing. CRISPR diagnostics have successfully detected various pathogens, including RNA viruses (eg.

The Pharma Data
JANUARY 20, 2021
Laboratory Implementation Laboratory reagents for performing the test commercially have been designed and are currently being validated in preparation for regulatory submission to CMS/CLIA with a view to targeting sales in the USA initially. MELBOURNE, Australia, Jan.

Roots Analysis
AUGUST 28, 2022
Furthermore, several live cell imaging and non-destructive fluorescence histological methods measure limited number of biomarkers at a time and the tags introduced may interfere with the natural function of genes and proteins. In fact, platform integration agreements account for 18% of the total deals inked in this domain, since 2016.

Drug Discovery World
FEBRUARY 27, 2023
PerkinElmer targets screening methods End-to-end solutions provider PerkinElmer launched two new products at the show targeting both high high-throughput screening (HTS) applications and next-generation screening (NGS). You always run the risk of missed expectations when you launch a follow-on product after a game changer like the LSA.

Drug Discovery World
FEBRUARY 15, 2023
3D organoid models provide more valuable information as they recapitulate both the structural and functional characteristics of a tumour, including glucose consumption, oxidative stress and lactic acid production. Reagents that can augment and enhance ADCC are of interest in addition to the antibodies themselves.
Drug Discovery World
JUNE 6, 2023
In our collateral, we have 56 genes, those are SMBs; we show that we can pick up those SMBs at the same quality if we were doing a separate alone DNA assay. A single product, quick workflow, getting you the answers as quickly as possible. It’s really all driven by the Archer technology.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content