Pilot and Feasibility Testing: The LIRE Example
Rethinking Clinical Trials
JULY 31, 2023
Jerry Jarvik, principal investigator of the LIRE Demonstration Project, discusses pilot and feasibility testing in pragmatic clinical trials.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

Rethinking Clinical Trials
JULY 31, 2023
Jerry Jarvik, principal investigator of the LIRE Demonstration Project, discusses pilot and feasibility testing in pragmatic clinical trials.

Pharma Times
DECEMBER 18, 2023
Earlier diagnosis could increase patient survival rates by five years or more The University of Glasgow has announced that an artificial intelligence (AI)-enhanced chest X-ray reporting solution has begun trials in NHS Greater Glasgow and Clyde (NHSGGC) for early detection of lung cancer.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

XTalks
NOVEMBER 7, 2022
Gleich, MD, FACS, Senior Vice President, Medical Department, and Dr. Christopher Huth, PhD, Senior Clinical Trial Manager, Clinical Trial Management. Liquid Biopsy Use in Oncology Clinical Trials. FoundationOne ® Liquid CDx (Foundation Medicine; end-to-end commercial provider). percent show less reliability).
The Pharma Data
MARCH 22, 2021
The AstraZeneca US Phase III trial of AZD1222 demonstrated statistically significant vaccine efficacy of 79% at preventing symptomatic COVID-19 and 100% efficacy at preventing severe disease and hospitalisation. The trial had a 2:1 randomisation of vaccine to placebo. The specific search for CVST found no events in this trial.

The Pharma Data
FEBRUARY 21, 2022
Our leadership in cardiology, radiology and women’s health is recognized worldwide, and we are expanding our presence in oncology, working tirelessly to bring forward new approaches that can change the treatment paradigm for patients,” said Stefan Oelrich, Member of the Board of Management, Bayer AG and President of Bayer’s Pharmaceuticals Division.
XTalks
MARCH 27, 2023
The first line of treatment for invasive candidiasis is a subset of antifungal medications called echinocandins, which includes caspofungin (brand name Cancidas), the drug that rezafungin was compared with in clinical trials. Once the fungus cannot synthesize β-(1, 3)-D-glucan, it undergoes osmotic instability and cell death.

Worldwide Clinical Trials
MAY 24, 2022
Join us as we celebrate cancer research awareness month and clinical trials awareness month. What I like about my role at Worldwide Clinical Trials is that feeling that I am close to the patient, as I am often involved in confirming the patient’s eligibility on the study, understanding how patients are responding to the drug.
Drug Discovery World
JANUARY 24, 2023
Virtual clinical trials ‘Virtual’ and ‘technology’ are concepts which are readily exchanged in this industry, but these are more than just buzzwords. The overall virtual clinical trials market is currently valued at $7.8 In early November 2022, two Alzheimer’s drugs were tested head-to-head in a first-ever virtual clinical trial 3.

Delveinsight
DECEMBER 29, 2020
Audentes Therapeutics’ ASPIRO Trial Free to Proceed. The Food and Drug Administration (FDA) decided to lift the clinical ban from Audentes Therapeutics’ ASPIRO trial, which was aimed at investigating its AT132 for the treatment of X-linked myotubular myopathy (XLMTM), a rare neuromuscular disease. . The post A USD 4.2

Scienmag
FEBRUARY 16, 2021
Food and Drug Administration (FDA)’s Center for Devices and Radiological Health (CDRH) will participate in this “collaborative community” A medical “collaboratory” is a forum in which multidisciplinary private and public sector members work together on medical […].
The Pharma Data
NOVEMBER 9, 2021
Still, the recommended cure for molnupiravir grounded on the Phase 3 MOVe- OUT clinical trial would be 800 mg doubly daily for five days, If authorized or approved.

pharmaphorum
SEPTEMBER 8, 2022
The Digital Medicine Society (DiME) is today releasing a new set of open-access resources to advance the use of nocturnal scratch in the treatment of atopic dermatitis (AD). . It is hoped today’s event will serve as a “blueprint for the broader development and deployment of digital endpoints in medical product development”.

Scienmag
SEPTEMBER 18, 2020
ARRS promises to deliver the same clinically relevant experience for which its Annual Meeting has long been heralded: world-class educational activities led by the field’s foremost experts that span every subspecialty of radiology Leesburg, VA, September 18, 2020–The American Roentgen Ray Society (ARRS) will convene its 2021 Annual Meeting (..)
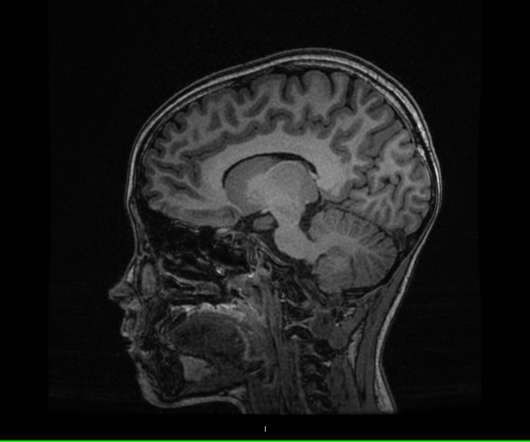
XTalks
NOVEMBER 29, 2022
Last week , researchers at the Yale School of Medicine reported that they have identified brain biomarkers of attention-deficit/hyperactivity disorder (ADHD) in children. The study results will be presented at the Radiological Society of North America annual meeting , which is currently being held in Chicago, IL until December 1, 2022.

The Pharma Data
MARCH 25, 2022
This includes about three million people in the EU who are immunocompromised or being treated with immunosuppressive medicines.(1). 2-4 Evusheld was generally well-tolerated in the trial.(2-4). 2-4 Evusheld was generally well-tolerated in the trial.(2-4). 2 Omicron SARS-CoV-2 subvariants in circulation around the world.(5-7)
XTalks
JANUARY 26, 2024
Personalized medicine: One of the most exciting prospects of CRISPR/Cas9 in oncology is its contribution to personalized medicine. Clinical trials have demonstrated the potential of CRISPR/Cas9 in this field. Clinical trials have demonstrated the potential of CRISPR/Cas9 in this field.

The Pharma Data
MARCH 24, 2021
Positive high-level results from the primary analysis of the Phase III trial of AZD1222 in the US have confirmed vaccine efficacy consistent with the pre-specified interim analysis announced on Monday 22 March 2021. The NIAID-supported COVID-19 Prevention Network (CoVPN) participated in the trial. D8110C00001 1.

Bioengineer
JULY 15, 2021
“In clinical trials, we tend to go with the maximum tolerable dose, the idea being that radiation kills the cancer and the more we give, the better. . “In clinical trials, we tend to go with the maximum tolerable dose, the idea being that radiation kills the cancer and the more we give, the better. ” ###.

The Pharma Data
MARCH 29, 2022
Evusheld significantly reduced the risk of developing symptomatic COVID-19 in PROVENT Phase III trial, with protection lasting at least six months Evusheld retains neutralising activity against the Omicron BA.2 1-3 Evusheld was generally well-tolerated in the trial. 2 subvariant, now the dominant strain in Europe. Christoph D.

Drug Discovery World
APRIL 8, 2024
Outstanding Achievement Award Pfizer Chairman and CEO Dr Albert Bourla received the 2024 AACR Outstanding Achievement Award for Service to Cancer Science and Medicine on behalf of Pfizer during the Opening Ceremony.

The Pharma Data
DECEMBER 23, 2020
Joseph Kim , President and CEO of INOVIO, said, “We are very pleased to share peer-reviewed Phase 1 clinical data for INO-4800 published in The Lancet’s EClinicalMedicine , and are grateful for the support of all participants and investigator staff involved in the clinical trial.” . mg and 2.0 mg and 2.0

The Pharma Data
MARCH 23, 2023
Darolutamide has already demonstrated efficacy and safety in nmCRPC with the Phase III ARAMIS trial, and in mHSPC with the Phase III ARASENS trial. The primary endpoint of this study is radiological progression-free survival (rPFS), measured by PSMA PET/CT assessed by independent central review.

XTalks
APRIL 27, 2021
Loftus, MD, acting director of the Office of Neurological and Physical Medicine Devices in the FDA’s Center for Devices and Radiological Health, in a statement from the agency. This included data from an unblinded study of 40 stroke patients over a 12-week trial.

The Pharma Data
OCTOBER 22, 2020
The approval of Veklury marks an important milestone in efforts to help address the pandemic by offering an effective treatment that helps patients recover faster and, in turn, helps preserve scarce healthcare resources,” said Barry Zingman, MD, Professor of Medicine at the Albert Einstein College of Medicine and Montefiore Medical Center, New York.

XTalks
AUGUST 30, 2022
Clinical research experts from the CRO Medpace shared insights about the operational and regulatory considerations for neuroscience trials with direct CNS administration. Why is it Difficult to Develop CNS Therapeutics?
pharmaphorum
SEPTEMBER 30, 2022
First and most obviously is the detailed data from the Clarity AD study reported this week, which will be revealed at the Clinical Trials on Alzheimer’s Congress (CTAD) in November. point advantage. “What we cannot know yet is whether that effect increases over time in an individual.

Drug Discovery World
APRIL 14, 2023
Carl H June, MD, FAACR – Lifetime Achievement in Cancer Research June, a Fellow of the AACR Academy, is the Richard W Vague Professor in Immunotherapy, Director of the Center for Cellular Immunotherapies, and Director of the Parker Institute for Cancer Immunotherapy in the Perelman School of Medicine at the University of Pennsylvania.

XTalks
MARCH 30, 2021
Loftus, MD, acting director of the Office of Neurological and Physical Medicine Devices in the FDA’s Center for Devices and Radiological Health, in a press announcement from the agency. The first study was a randomized, double-blind, controlled trial consisting of 20 MS patients with gait deficits.
The Pharma Data
NOVEMBER 19, 2020
At this point, the procedure is slowly becoming a more established treatment, said Dr. Felix Gonzalez, a radiologist at Emory University School of Medicine in Atlanta. And before the ablation is done, Gonzalez explained, patients go through what is basically a trial run. 29 to Dec.

pharmaphorum
APRIL 7, 2021
billion-plus alliance with UK biotech Artios Pharma to develop precision medicines for cancer. Meanwhile, it also has a candidate in late-stage testing, called Lu-PSMA-617, which improved survival in PSMA-positive castration-resistant prostate cancer (CRPC) in a phase 3 trial last month and has been billed as a future blockbuster.

The Pharma Data
AUGUST 15, 2021
The approval is based on results from the open-label Study 004 trial (N=61), where the major efficacy endpoint was overall response rate (ORR) in patients with VHL-associated RCC. The major efficacy endpoint for the treatment of VHL-associated RCC was ORR measured by radiology assessment using RECIST v1.1 WELIREG is the first HIF-2?

The Pharma Data
NOVEMBER 6, 2020
director of the Office of Neurological and Physical Medicine Devices in the FDA’s Center for Devices and Radiological Health. This device was studied in a 30-day randomized, sham-controlled trial of 70 patients. Nightware is available by prescription only and is intended for home use.

The Pharma Data
NOVEMBER 18, 2021
Six-months follow-up of prevention trial showed 83% reduced risk of symptomatic COVID-19, with no severe disease or deaths with AZD7442. Separate treatment trial showed 88% reduced risk of severe COVID-19 or death when treated within three days of symptom onset. In both PROVENT and Paraphernalia, AZD7442 was generally well permitted.

The Pharma Data
OCTOBER 12, 2021
Trial met primary endpoint. The trial met the primary endpoint, with a cure of 600 mg of AZD7442 given by intramuscular (IM) injection reducing the threat of developing severe COVID-19 or death (from any cause) by 50 compared to placebo in rehabilitants who had been characteristic for seven days or lower.
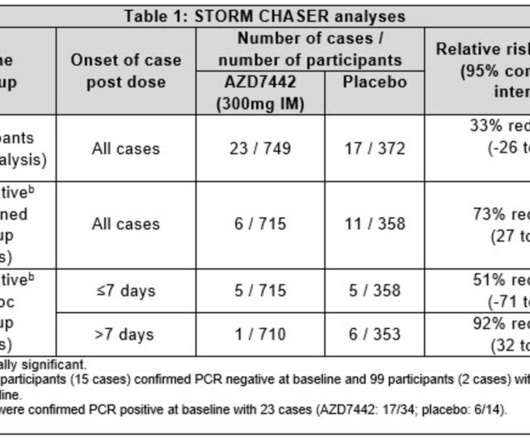
The Pharma Data
JUNE 25, 2021
AstraZeneca today announced results from the STORM CHASER trial assessing the safety and efficacy of AZD7442, a long-acting antibody (LAAB) combination, for the prevention of symptomatic COVID-19 in participants recently exposed to the SARS-CoV-2 virus. The PROVENT trial will give us more clarity in this patient population.

Drug Discovery World
APRIL 16, 2023
DDW’s Megan Thomas rounds up highlights from Sunday 16 April at AACR 2023, from promising trial results for a personalised mRNA-based cancer vaccine to winners of lifetime achievement awards.
The Pharma Data
AUGUST 22, 2021
The trial accrued 25 cases of symptomatic COVID-19 at the primary analysis.There were no cases of severe COVID-19 or COVID-19-related deaths in those treated with AZD7442. The trial included 5,197 participants in a 2:1 randomisation AZD7442 to placebo. The trial was conducted in 87 sites in the US, UK, Spain, France and Belgium.

The Pharma Data
DECEMBER 8, 2020
Under the Orphan Drug Act, FDA may provide grant funding toward clinical trial costs, tax advantages, FDA user-fee benefits, and seven years of market exclusivity in the United States following marketing approval by FDA. Currently, there is no cure for IPF and only two drugs are approved in the United States to treat the condition.

The Pharma Data
MARCH 17, 2021
The data builds on the primary outcome analysis of the positive Phase 3 monarchE trial that previously showed Verzenio, in combination with ET, decreased the risk of breast cancer recurrence by 28.7 The monarchE trial is ongoing and patients will continue to be followed to assess safety, PROs and other endpoints. had fatal outcomes.
XTalks
JANUARY 19, 2024
Radiobiological and Clinical Considerations from CRO and Emerging Biotech Radiopharmaceuticals are medicinal formulations containing radioisotopes that are used in the field of nuclear medicine primarily for the diagnosis and treatment of various diseases, particularly cancer. There have been more than $1.7
Delveinsight
DECEMBER 10, 2020
The full data from late-phase trials of AstraZeneca’s COVID-19 vaccine have pointed to the potential for AZD1222 to work after a single dose and limit asymptomatic cases; however, missed main questions regarding the efficacy of the candidate. In a pooled analysis of 11,636 participants enrolled in two separate clinical trials in the U.K.

The Pharma Data
APRIL 7, 2023
” These sNDAs are supported by results from the PHAROS trial (NCT03915951), an open-label, multicenter, non-randomized, Phase 2 study (n=98) to determine the safety, tolerability, and efficacy of BRAFTOVI given in combination with MEKTOVI in patients with BRAF V600E-mutant metastatic NSCLC.

The Pharma Data
NOVEMBER 18, 2020
. “Effective delivery of this treatment can be logistically complicated,” said Dr. Kami Kim , director of the Division Infectious Diseases and International Medicine at USF Health Morsani College of Medicine. ” The monoclonal antibody is a man-made protein that acts like a human antibody in the immune system.

The Pharma Data
DECEMBER 9, 2020
. “Of all the organs in our body, the brain is the one most easily damaged by a poor diet,” said Dr. Lisa Mosconi, director of the Women’s Brain Initiative and an associate professor of neuroscience in neurology and radiology at Weill Cornell Medicine and NewYork-Presbyterian Hospital. So, what does work?
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content