FDA puts breakthrough tag on Roche, Lilly Alzheimer’s test
pharmaphorum
APRIL 11, 2024
Roche and Eli Lilly have been granted breakthrough status by the FDA for their pTau217 blood test for Alzheimer's disease.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

 tag testing
tag testing 
pharmaphorum
APRIL 11, 2024
Roche and Eli Lilly have been granted breakthrough status by the FDA for their pTau217 blood test for Alzheimer's disease.

Medical Xpress
MARCH 8, 2023
Before new therapies can reach patients, they must be tested in clinical trials in representative populations to show that they work and are safe. Failure to enroll enough participants in trials can delay the arrival of new therapies in the clinic and inflate their eventual price tags.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.
STAT News
AUGUST 16, 2022
At issue is a TB test developed by Cepheid, which reached a 10-year deal with the WHO in August 2012 to supply its GeneXpert diagnostic to dozens of low- and middle-income countries. million, which was provided by the WHO and others, the company agreed to supply its test for $9.98 In exchange for $11.1

Rethinking Clinical Trials
OCTOBER 26, 2023
point of care diagnostic tests, provide broad access to curative Hepatitis C medications with a national subscription model, with Medicare co-pay assistance and commercial insurance coverage. PCORnet is engaging with sites to support the ITAP clinical study for de novo clearance of a qualitative POC HCV test-to-treat platform.
XTalks
APRIL 18, 2024
Roche has received Breakthrough Device designation from the US Food and Drug Administration (FDA) for its Elecsys pTau217 early Alzheimer’s blood test. This test, which is being co-developed with Eli Lilly, has the potential to facilitate earlier and more accurate detection of the disease.

XTalks
MAY 2, 2024
During a midday session, Anal Dave from Dessert Holdings and David Hatch, Vice President of Digital Solutions Marketing at Neogen , showcased their digital transformation strategies in environmental testing, enhancing detection and compliance. improved strain discrimination in testing, reducing both time and cost.

Rethinking Clinical Trials
JULY 22, 2022
We selected 35 and completed testing of 27 of them. Five agents are still in testing. Having the companies at the table, bringing the resources (man power, willingness to test, openness of conversation) was amazing and contributed to success of the program. The team reviewed 800 candidates over 2 years time. Discussion Themes.

Rethinking Clinical Trials
APRIL 18, 2023
This broad set of clinical conditions and varied underlying causes underscore the need for testing a broad portfolio of therapeutic agents. RECOVER is a patient-centered, integrated, adaptive research network. It seems the most severely affected people came from the early waves of COVID.

Rethinking Clinical Trials
FEBRUARY 9, 2023
This trial tested the hypothesis that the use of a platform-agnostic, EHR-embedded VTE risk model with integrated CDS would 1) increase rates of appropriate Thromboprophylaxis, and subsequently 2) reduce thromboembolism, compared to usual medical care in hospitalized, medically-ill patients.

Rethinking Clinical Trials
JULY 27, 2022
ACTIV-6 is testing medication doses approved by the FDA for other purposes, i.e., repurposed drugs. Participants test positive for COVID-19 with a FDA-authorized test, register from home, are randomized remotely, receive study medication through the central pharmacy, and follow instruction until their clinical symptoms improve.

Rethinking Clinical Trials
JULY 13, 2023
Discussion Themes – How diverse were the settings where this was tested? The study team was interested in testing in a setting outside of a large academic hospital. We need to test in systems that might have an enhanced usual care arm. Learn more Read about the IMPLEMENT-HF study in JACC.

Rethinking Clinical Trials
FEBRUARY 14, 2024
The preparation work started with an idea in 2010, which led to selection of the pilot study in 2014 to test out the system for randomizing patients within the existing infrastructure. How long did the start up for the trial take? The actual startup was about 18 months.

The Pharma Data
NOVEMBER 29, 2021
1.1.529 a variant of concern, named Omicron, on the advice of WHO’s Technical Advisory Group on Virus Evolution (TAG-VE). The number of people testing positive has risen in areas of South Africa affected by this variant, but epidemiologic studies are underway to understand if it is because of Omicron or other factors.

Rethinking Clinical Trials
AUGUST 22, 2022
Trial participants were adults ages 30-85, who tested positive for SARS-CoV-2 within 3 days, with fewer than 7 days of symptoms, no known prior infection with SARS-CoV-2, no severe kidney, liver, or heart failure. We used Google Ads and partnerships with some testing clinics. We also kept equal focus on follow-up.

Drug Discovery World
AUGUST 15, 2022
She is a Senior Enterprise Fellow at the University of Southampton and coordinates the AI 4 Scientific Discovery Network and the Future Blood Testing Network, in addition to running an Ethics working group.

Rethinking Clinical Trials
SEPTEMBER 22, 2022
The DANFLU-1 trial is a large-scale pragmatic RCT that looked at high-dose vs standard-dose flu vaccine to test whether high-dose vaccines might reduce hospitalizations among the elderly, age 65 and older. The aim is to test communication strategies for optimizing the update of the flu vaccine. Learn more. Read about DANFLU-1.
Drug Discovery World
JANUARY 9, 2023
In this early research, Khalid Shah, Professor of Neurosurgery at Harvard Medical School and Brigham and Women’s Hospital, and colleagues tested the vaccine in an advanced mouse model of brain cancer glioblastoma, with promising results. The findings are published in Science Translational Medicine. .

Rethinking Clinical Trials
MAY 25, 2023
Researchers aimed to test the safety and effectiveness of Aspirin use as prophylaxis in orthopedic trauma cases. Tags #pctGR, @Collaboratory1 The post Grand Rounds May 19, 2023: Aspirin or Low-Molecular-Weight Heparin for Thromboprophylaxis After a Fracture (Robert O’Toole, MD) appeared first on Rethinking Clinical Trials.

Rethinking Clinical Trials
JANUARY 26, 2023
Tags #pctGR, @Collaboratory1 The post Grand Rounds January 20, 2023: Collaborative Pragmatic Trials in Action: EVOLVE-MI (Mikhail Kosiborod, MD) appeared first on Rethinking Clinical Trials.

Rethinking Clinical Trials
JULY 19, 2023
The objective of COORDINATE-Diabetes trial was to improve the implementation and adoption of these therapies by testing the impact of a clinic-level, multifaceted intervention on the prescription of 3 key groups of evidence-based therapies. However, these therapies are highly underused in routine clinical practice. Granger, MD; Neha J.
Delveinsight
FEBRUARY 23, 2021
The company plans to use the capital to expand its pipeline of protein-based vaccines and biologic cancer therapies utilizing its innovative and proprietary Trimer-Tag technology platform. M from BARDA for SARS-CoV-2 Antigen Test. Moreover, the test is set to deliver the results in less than 6 minutes.

Rethinking Clinical Trials
AUGUST 30, 2023
The RA PRO dashboard was tested as part of a stepped wedge, cluster randomized trial at the clinician level. The team collected focus group feedback from patients and clinicians and worked with IT professionals to pull data directly from the electronic health record and design an interface that could be used by all patients.

Rethinking Clinical Trials
OCTOBER 11, 2023
Through this initiative, 7 Clinical Centers are expected to test an evidence-based, multi-level intervention designed to reduce or eliminate cardiovascular and/or pulmonary health disparities. Tags #pctGR, @Collaboratory1 The post Grand Rounds October 6, 2023: Hybrid Studies Should Not Sacrifice Rigorous Methods (David M.

Drug Discovery World
NOVEMBER 7, 2022
Cereblon, the researchers determined, has a default ‘open’ conformation, but must be switched to a particular ‘closed’ conformation for the ubiquitin-tagging of target proteins. . So now they know, and they can test their drugs for this key property.” .

pharmaphorum
SEPTEMBER 15, 2020
“Digitally native brands are typically agile businesses from the ground up, and agility and flexibility are part of their core makeup,” says Joe Holmes, vice president, marketing services, US at Tag. They also employ exceptionally high levels of ‘test and learn’ activities to understand which campaigns work and should be upscaled.

pharmaphorum
JANUARY 11, 2022
It has since been joined by other products in the software as a medical device (SaaMD) category, including Pear Therapeutics’ alcohol-use disorder candidate Pear-009 and another Blue Note DTx, codenamed BNT001, which is also being tested for anxiety and depression in cancer patients.

Rethinking Clinical Trials
AUGUST 28, 2023
Its objective is to improve efficiency of new drug development and registration processes, in addition to promoting public health, preventing duplication of clinical trial in humans and minimizing the use of animal testing without compromising safety and effectiveness. The guidance should not remove the need to think.

Rethinking Clinical Trials
AUGUST 28, 2023
Its objective is to improve efficiency of new drug development and registration processes, in addition to promoting public health, preventing duplication of clinical trial in humans and minimizing the use of animal testing without compromising safety and effectiveness. The guidance should not remove the need to think.

pharmaphorum
DECEMBER 23, 2021
The Japanese drugmaker was originally developing patritumab as an antibody for NSCLC, but abandoned the drug after it showed little efficacy in mid-stage testing, pivoting to the ADC version. The post FDA gives Daiichi Sankyo’s HER3 drug a breakthrough tag appeared first on.
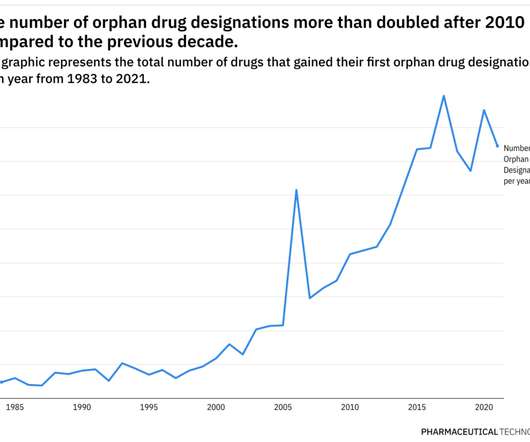
Pharmaceutical Technology
JUNE 29, 2022
For example, while orphan drug designations have more than doubled in the last decade compared to the previous one, only 16% of therapies with orphan tags have managed to gain FDA approval in some indications. Prior to the program, only 10 drugs were approved for a rare disease.

pharmaphorum
DECEMBER 12, 2022
Vyvgart is already approved to treat generalised myasthenia gravis, getting a green light for that indication from the FDA a year ago, and launched with a $225,000 per annum price tag in competition with rival gMG drugs like AstraZeneca/Alexion’s C5 inhibitor Soliris (eculizumab).

pharmaphorum
NOVEMBER 19, 2020
Because Roche has never applied for Avastin to be used in wet AMD, clinics in the US have had to choose between the peace of mind offered by the regulatory process and Lucentis’ higher price tag, or the unapproved off-label formulation of Avastin at a lower price.

The Pharma Data
MARCH 23, 2022
Back in 2018, researchers tested a broad-spectrum antiviral candidate called remdesivir/VEKLURY, which acts as a nucleotide decoy to get incorporated into the viral RNA genome and stop viral polymerase. To test this, the LJI team partnered with their long-term collaborator, the UTMB research group led by Professor Alexander Bukreyev, Ph.D.,

pharmaphorum
MARCH 23, 2022
Vyvgart (efgartigimod alfa) became the first drug in the FcRn class to be approved by the FDA last December, and despite a $225,000 annual price tag is predicted to make relatively modest sales in its IV version, which will compete with other IV gMG drugs like AstraZeneca/Alexion’s C5 inhibitor Soliris (eculizumab).

The Pharma Data
DECEMBER 3, 2020
Preliminary results from stability studies have demonstrated that S-Trimer is stable at 2-8 o C for at least six months (longer-term stability studies are ongoing) and stable at room temperature and 40 o C for at least one month, in line with the adjuvants tested. About Trimer-Tag © Technology. About COVID-19 S-Trimer Vaccine.

XTalks
JANUARY 28, 2022
Kimmtrak has a price tag of $18,760 per vial, which amounts to a weekly dose. Related: Cancer Blood Test Developed by University of Oxford Researchers Can Detect Metastasis. The company is expecting approval from the European Medicines Agency (EMA) soon as it prepares for a European launch in the second quarter of this year.
Drug Discovery World
JANUARY 10, 2023
Bethyl antibodies are highly regarded for passing strict validation testing before arriving in customer hands. Pillar 5: Protein OE/Epitope Tags. We generate numerous candidates for you to test in your assays and workflows ensuring the antibodies are effective in whatever application they’re being used.

pharmaphorum
JANUARY 5, 2021
TIGIT is very much among the front runners however, and Roche is being closely pursued by other drug developers including Merck & Co with MK-7684 and Gilead/Arcus, which are also in mid-stage testing, as well as iTeos Therapeutics and Mereo BioPharma.

XTalks
JANUARY 7, 2022
Throughout the month, guests who opt for a noodle swap of LEANguini, Zoodles or Caulifloodles at the test locations will receive double the Noodles Rewards points on their order. Even LEANguini comes at a higher price tag since it commands an $8 to $10 surcharge when substituted in any made-to-order dish.

pharmaphorum
OCTOBER 29, 2021
Rival drug Tecentriq (atezolizumab) – had its accelerated approval in first-line TNBC withdrawn by developer Roche in the US after failing a confirmatory trial, but is in late-stage testing as a treatment for early-stage TNBC. The post Gilead tags Merck for first-line triple negative breast cancer trial appeared first on.

Delveinsight
FEBRUARY 25, 2021
GRAIL and Quest collaborate for the cancer blood test. Cancer detection company GRAIL has announced a collaboration with Quest Diagnostics that sees Quest proffering phlebotomy services to bolster GRAIL’s multi-cancer early detection blood test Galleri. once Galleri becomes accessible in the second quarter of 2021.
pharmaphorum
DECEMBER 23, 2022
price tag of $475,000 when it was first launched in 2017 – and it becomes apparent that these may not be desirable treatment options for every patient and in every setting. Add to this the considerable cost of these medications – the first approved CAR-T, Novartis’ Kymriah (tisagenlecleucel), had a U.S. CellCentric, a U.K-based

Rethinking Clinical Trials
JANUARY 23, 2024
Feasibility and generalizability affect design choices including inclusion/exclusion criteria, flexibility of imaging intervention being tested, guidance/control of subsequent care after imaging, and endpoints and outcomes. When we evaluate imaging strategies for chest pain, is “usual testing” the appropriate comparator?

Intouch Solutions
APRIL 11, 2024
It has continually evolved, embracing new technologies and methodologies to improve drug discovery, development, and distribution. This iterative process yields several benefits, including: Creating clusters of customers based on actual physician behavior.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?

Let's personalize your content