bluebird bio wins back-to-back landmark FDA approvals for first-in-class gene therapies
Pharmaceutical Technology
SEPTEMBER 30, 2022
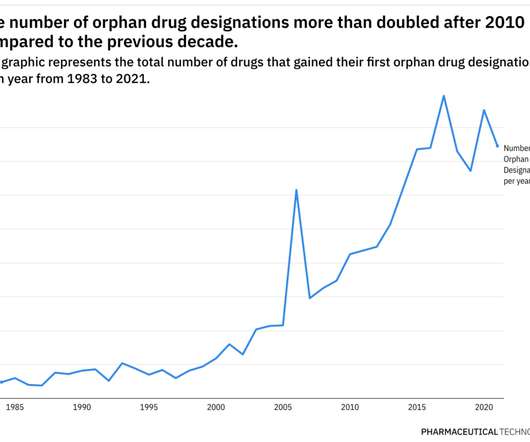
Spark Therapeutics’ Luxturna, indicated for inherited retinal disease (IRD), was the first gene therapy to be approved, in 2017, with a price tag of $850,000 for each eye. This highlights the FDA’s dedication to pursuing innovative, life-saving therapies for patients without any alternatives.













































Let's personalize your content